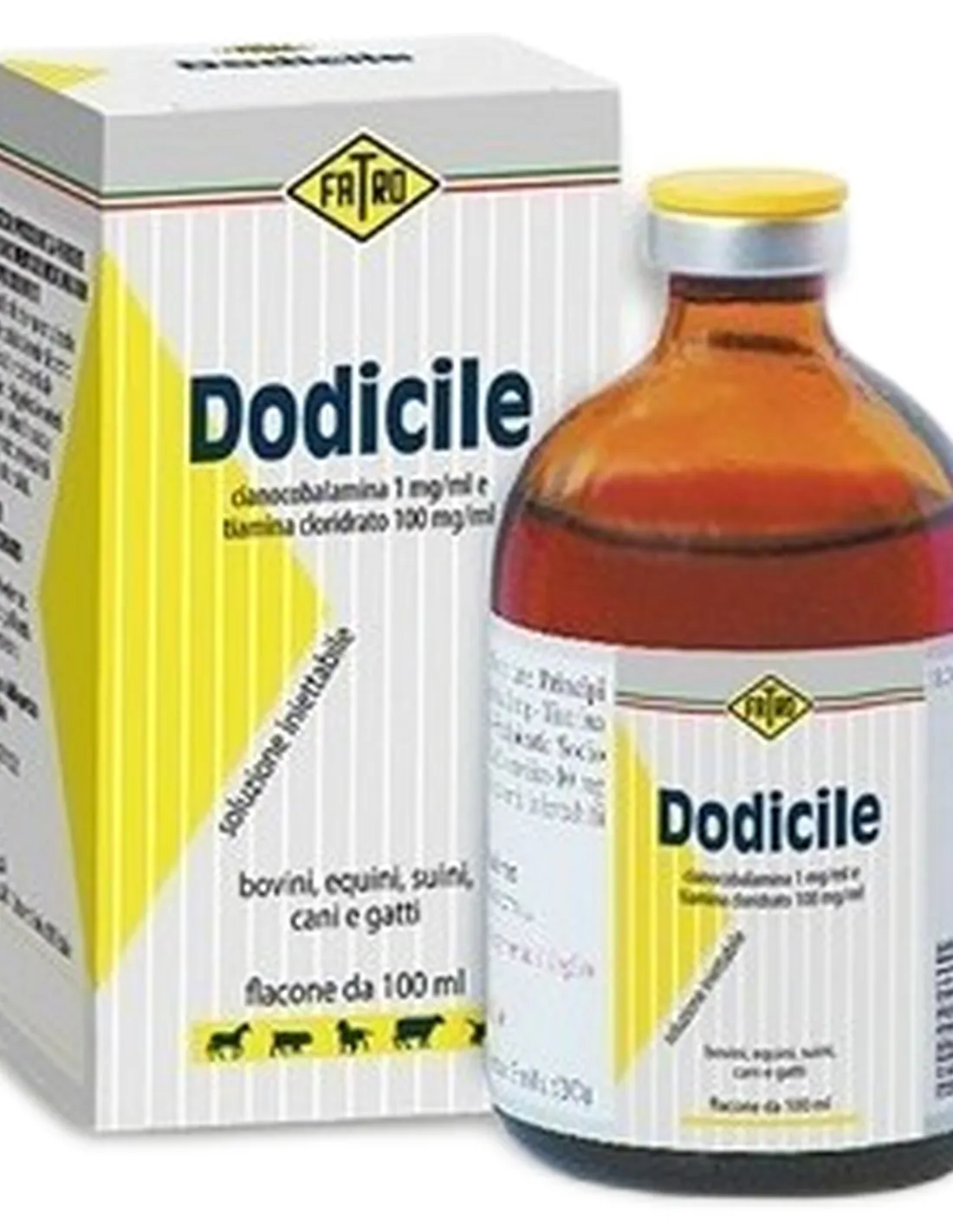
PHARMACOTHERAPEUTIC CATEGORY: Vitamins
COMPOSITION:
Active ingredients: Cyanocobalamin 1 mg/ml; thiamine 89.2 mg/ml equal to thiamine hydrochloride 100 mg/ml.
Excipients: Benzyl alcohol; disodium edetate; water for injectable preparations.
INDICATIONS:
It is indicated in cases of: hepatofunctional and metabolic disorders (ketosis, liver disease, toxic states); growth disorders and states of organic deterioration (anorexia, asthenia, convalescence); nervous and neuromuscular, deficiency and toxic disorders (neuritis, neuralgia, paresis); states of toxic, infectious, parasitic anemia, blood dyscrasias; states of muscle fatigue.
CONTRAINDICATIONS/SIDE EFFECTS:
Do not use in case of known hypersensitivity to the active ingredients or to any of the excipients.
PHARMACEUTICAL FORM: Injectable solution
USE/ROUTE OF ADMINISTRATION: Intramuscular, intravenous.
DOSAGE:
Cattle, equines: 5-10 ml/head.
Calfs, foals: 2.5-5 ml/head.
Pigs: 1.5-4 ml/head.
Piglets: 1-1.5 ml/head.
Dogs, cats: 0.5-1.5 ml/head.
Possibly repeat every 5-7 days.
STORAGE:
Keep away from light and heat sources. Shelf life after first opening the container: 28 days.
WARNINGS:
Respect normal aseptic precautions. Special precautions for those administering the product: be careful when administering the product to avoid accidental self-injections. People with known hypersensitivity to thiamine or B vitamins should avoid contact with the veterinary medicinal product. Incompatibility: do not mix with other veterinary medicines.
TARGET SPECIES:
Cattle, horses, pigs, dogs, cats.
WITHDRAWAL PERIOD: Meat: zero days. Milk: zero hours.
INTERACTIONS:
Carbonates and citrates can reduce the effect of thiamine. Vitamin B12 may interact with chloramphenicol. The administration of antibiotics can distort the concentrations of vitamin B12.
DIAGNOSIS AND PRESCRIPTION:
Veterinary medicine without the obligation of a veterinary prescription.
SIDE EFFECTS:
None reported.
PREGNANCY AND BREASTFEEDING:
Can be used during pregnancy and breastfeeding.