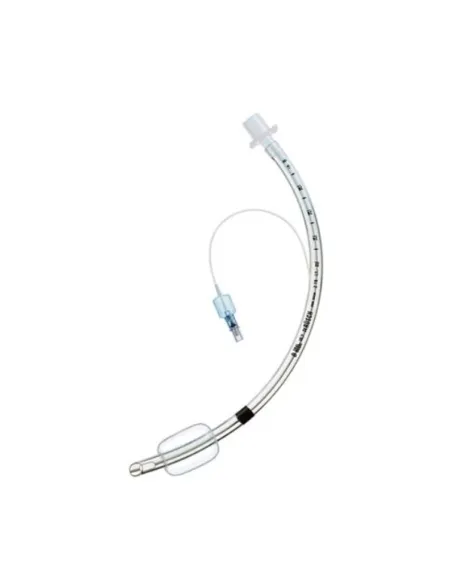
The sterile cuffed 9 mm tracheotube represents a fundamental solution for airway management in patients who require mechanical ventilation or who have upper airway obstruction. Specifically designed to guarantee safety, comfort and ease of use, this medical device stands out for the quality of the materials used and for its construction precision. The presence of the cap allows you to effectively isolate the respiratory tract, preventing the risk of aspiration of secretions or liquids and thus improving the control of gas exchange. The use of sterile packaging guarantees the maximum level of safety in application, reducing the risk of cross-infection and making it ideal for emergency procedures as well as for structured hospital settings. The internal diameter of 9 mm is suitable for adults or patients who require tracheal support of this caliber, ensuring optimal air passage even for long periods of use. Each package contains only one piece, for safe single use and compliant with modern clinical needs.
The ergonomic design of the Tracheotube allows for easy insertion and removal, while the smooth surface minimizes trauma to the tracheal mucosa during introduction. The tube maintains adequate flexibility to follow the natural morphology of the trachea without compromising patency. The high volume, low pressure cuff safeguards the tracheal tissues, allowing an effective seal without creating long-term damage to the tracheal wall.
Therapeutic Indications
The sterile cuffed 9 mm tracheotube finds its main indication in all those patients who require advanced airway management, both in emergency settings and in elective settings. It is particularly indicated in cases of respiratory failure requiring invasive ventilatory support, during surgical operations under general anesthesia, for the protection of the respiratory tract in patients at risk of aspiration and in trauma with compromise of the upper airway. It can also be used for the removal of tracheobronchial secretions, facilitating regular cleaning of the airways in the context of intensive monitoring.
The integrated cuff allows an optimal tracheal seal even in agitated patients or in the presence of high ventilatory pressures, effectively preventing the passage of unwanted materials into the lower airways. The product proves to be very useful not only in emergency rooms and intensive care departments, but also in pre-hospital emergency medicine and during intra- or inter-hospital transfers, always guaranteeing a safe and stable airway. Furthermore, it finds application in patients with laryngeal edema, vocal cord paralysis or airway tumors, where early and controlled access to the trachea is essential for survival.
Finally, it is also indicated for temporary respiratory support in situations of deep sedation, coma or neuromuscular block, ensuring simple and safe management of anesthetic gases and the prevention of inhalation complications.
Composition
The sterile cuffed 9 mm tracheotube is made with highly biocompatible medical grade materials, chosen to offer safety, flexibility and resistance. The main structure of the tube is made of transparent medical PVC (polyvinyl chloride), which guarantees maximum internal visibility and allows monitoring of any secretions or residues present inside the lumen. This feature is particularly important for healthcare personnel in order to prevent blockages and promote optimal maintenance during prolonged use.
The cuff positioned at the distal end is made of soft and resistant material, inflatable using a special control balloon, allowing both monitoring of the cuff pressure and manual adjustment of the volume according to the patient's needs. All materials are latex-free, so as to prevent possible allergic reactions in predisposed subjects. Integrated accessories include universal connectors for connection to mechanical ventilation circuits or suction systems, as well as graduated depth indicators on the surface of the tube to facilitate precise and correct tracheal position.
The sterile packaging in a single pouch ensures total protection of the device until the moment of use and avoids the risk of contamination. All components are certified for use in hospitals and meet the quality and safety standards required by current legislation on disposable medical devices.
Contraindications
The sterile cuffed 9 mm tracheotube should not be used in patients with obvious anatomical alterations of the airway that preclude safe insertion of the tube, or where the 9 mm size may not be compatible with the patient's tracheal caliber (for example in pediatric patients or with a smaller diameter trachea). It is also contraindicated in subjects with known or suspected hypersensitivity to the constituent materials, including PVC or silicone components used in the production of the cap.
It is not indicated for non-invasive ventilation procedures or for intubations to assist oxygen therapy alone without the need for tracheal sealing. It must be used with particular caution in patients with coagulation disorders or tracheal fragility, due to the risk of bleeding or injuries due to insertion or prolonged presence of the device. Prolonged or inappropriate use of the tracheotube can expose the patient to complications, including tracheal stenosis, compression injuries of the cuff or local infections.
Other potential risks to consider include accidental dislodgement, occlusion of the tube by thick secretions, and difficulty in removal in the presence of pronounced lower airway edema. For these reasons, the device must only be used by qualified healthcare personnel and with careful evaluation of individual clinical conditions.
Safety of the drug for the species
In the context of human use, the safety of the cuffed 9mm Tracheotube has been extensively documented through standardized airway management protocols. The low pressure cuff reduces the risk of tracheal injury even in the event of prolonged use, provided that the internal pressure is constantly monitored to avoid ischemic damage from compression. The transparency of the tube allows a constant evaluation of the secretions, guaranteeing timely interventions in case of obstruction.
The use of biocompatible and disposable materials eliminates the possibility of infectious transmission from patient to patient, while the absolute sterility of the packaging represents a guarantee in all conditions of use, including departments with high infectious risk. The risk of allergic reactions is extremely reduced thanks to the total absence of latex or plasticizers with a high risk of sensitization. The design of the tracheotube also provides for the absence of sharp edges or parts, reducing the risk of accidental damage to the tracheal mucosa during insertion or removal.
However, it is essential to adopt constant monitoring of the positioning and conditions of the tube throughout the period of use, to prevent complications such as accidental decannulation, occlusion by mucus or bacterial colonization of the device itself. Correct staff training and the use of aseptic techniques during handling represent the best strategies to optimize patient safety.
Precautions
Before use, always check the integrity of the packaging and the correct functioning of the inflatable cap, which must maintain pressure without obvious leaks. The procedures for insertion, management and removal of the sterile cuffed 9 mm tracheotube must be conducted by appropriately trained professionals, in a suitable environment and adopting all the aseptic measures necessary to prevent local or systemic infections.
The cuff pressure must be carefully monitored using a suitable manometer, avoiding excessive pressures which could cause ischemia and necrosis of the tracheal wall. In case of prolonged use, carry out regular checks of the patency of the tube and the presence of secretions, encouraging their removal through safe suction techniques. Never force insertion if there is resistance, to avoid trauma or injury to the laryngeal and tracheal structures.
Pay particular attention to patients with previous tracheal pathologies, inflammatory processes or ongoing infections, and to those who are particularly susceptible to complications from foreign bodies in the airways. The device, being disposable, must be disposed of according to medical waste guidelines once removed, without attempts at reuse or secondary sterilization.
Method of administration
The insertion of the sterile cuffed 9 mm tracheotube is typically performed under visual guidance, using a laryngoscope or flexible bronchoscope, in adequately anesthetized or sedated patients. Once the correct introduction through the vocal cords and the central position in the trachea has been verified, the cuff is inflated using the pilot balloon until an effective seal is obtained, constantly monitoring the optimal pressure indicated by the clinical guidelines.
The tube must be adequately fixed with special devices to prevent accidental movements and must be connected to assisted ventilation systems, monitoring the functionality with clinical evaluation and diagnostic aids (capnography, auscultation, radiography). While the tracheotube is in place, periodic secretion suction maneuvers and regular assessments of the state of the mucous membranes and cuff pressure must be guaranteed. Removal must be planned safely, with alternative ventilatory support if necessary.
Treatment program
The use of the sterile cuffed 9 mm tracheotube is typically configured as a temporary solution for airway management, although it can be kept in place for prolonged periods based on the clinical need of the individual patient. The duration of treatment is variable and depends on the specific indication: it can last a few hours during surgery or several days or weeks in the presence of the need for prolonged ventilation.
The monitoring program includes frequent checks of the position of the tube, the integrity of the cuff and the pressure inside it, with adjustments aimed at avoiding both leaks and damage from prolonged compression. In the event of obstruction or loss of function, the device must be replaced immediately to ensure adequate airway patency. Regular aspiration maneuvers and preventive actions against infections must be foreseen, including optimal management of fixation devices and connections with ventilation circuits. The personnel in charge must be constantly updated on the best practices for the management of cuffed tracheotubes, particularly in complex areas such as intensive care or emergency medicine.
Target species
Can be used exclusively for human patients who require advanced management of the airway, in all age groups in which the 9 mm caliber is compatible with the tracheal conformation. Do not use in pediatric patients or in subjects with a trachea of insufficient diameter for this size, to avoid the risk of injury, obstruction or permanent damage to the airway structures.