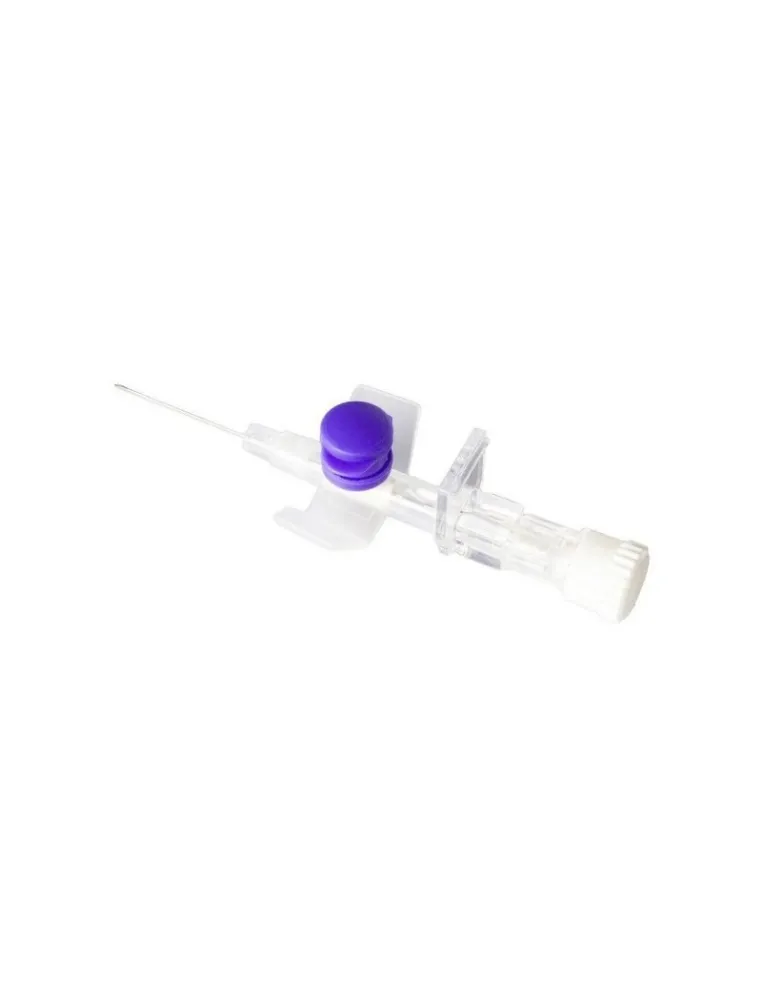
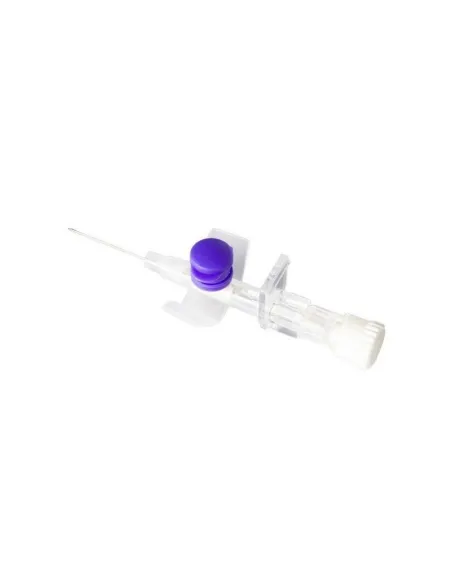
Delta Ven 2 G26 19MM is a medical device designed to offer an effective and reliable solution in peripheral venous access, ideal for contrast media procedures, blood sampling and drug infusion. Thanks to its configuration with a G26 needle and a length of 19 mm, it allows optimal management of venous access in both adults and pediatric patients, ensuring comfort and safety. The smooth surface and precise dimensions of the needle facilitate insertion, reducing the risk of trauma and improving adherence inside the blood vessel. The system is equipped with safety devices to minimize the risk of accidental punctures even during handling after use. The product is sterile, disposable and free of elements that could cause adverse reactions for the patient.
Designed for compatibility with different infusion systems, Delta Ven 2 G26 19MM ensures maximum reliability thanks to the high quality materials used in its production. The catheter, made of a flexible and hypoallergenic material, adapts perfectly to peripheral veins, facilitating both short infusions and prolonged treatments. The sharp tip needle guarantees effective penetration, reducing the sensation of pain for the patient and the risk of local complications such as bruising or irritation.
The product is individually packaged in guaranteed sterile conditions, with an easy opening system and without latex, to meet the needs of even the most sensitive patients. Each device has a transparent identifying color which facilitates immediate identification of the measurement and allows rapid recognition during multidisciplinary procedures.
Composition
Main materials: The catheter is made of the latest generation flexible medical material, completely free of latex and PVC, minimizing any risk of allergies or irritation for both the patient and healthcare personnel. The internal and external surface of the catheter is ultra-smooth, to ensure maximum smoothness during positioning in the vein and maintain a constant seal throughout the procedure.
The tip of the needle, with a G26 diameter and 19 mm long, is made of biocompatible stainless steel, equipped with anti-corrosion treatment and a sharp tip to minimize the risk of breakage, bending or vascular injury.
The product includes a self-sealing membrane that reduces blood leakage and a transparent shiny section that allows immediate visualization of blood reflux to verify correct positioning. There are additional safety components: protective cap, ergonomic non-slip base, universal Luer-Lock fitting for ease of connection to syringes and infusion sets. No part of the product contains phthalates, DEHP or other harmful plasticizers.
Contraindications
The use of Delta Ven 2 G26 19MM is contraindicated in the presence of active skin infections on the chosen insertion site, in patients with a known allergy to the materials that make up the needle or catheter, or in subjects suffering from uncontrolled coagulation disorders. Application is not recommended on thrombosed, sclerotic, inflamed or already damaged veins from previous catheterizations.
Do not use in patients with histories of anaphylactic reactions to vascular devices or in case of incompatibility with procedures required by specific therapy, such as infusions of highly viscous solutions or irritants to small peripheral veins. Avoid use in the same venous site as other equipment temporarily kept in situ.
The product is not intended for arterial use or central access. If pain, redness or fluid leakage occurs from the insertion site, stop use immediately and consult a doctor.
Precautions
Before use, make sure that the packaging is intact and has no holes or traces of contamination. Handle the product only after performing thorough hand hygiene and, where indicated, using sterile gloves. Insertion must be carried out exclusively by trained personnel following asepsis protocols for the prevention of infections at the insertion site.
Store the device in a cool, dry place, away from heat sources, direct sunlight and excessive humidity. Do not reuse: the Delta Ven 2 G26 19MM is disposable and reuse carries serious risks of infectious transmission. After use, dispose of the device according to local regulations regarding infectious medical waste.
In case of difficulty with insertion, blood leakage, swelling or any other anomaly, immediately suspend the maneuver and use a new device. Frequently monitor the state of the insertion site to prevent complications such as phlebitis, infiltration or risks of drug extravasation.