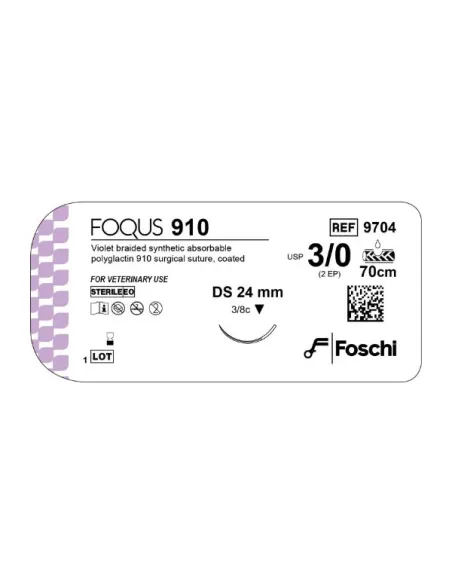
Foqus USP 1 Purple Circular Needle is a type of surgical needle designed to offer precision and reliability in soft tissue suturing procedures. This needle, characterized by its circular shape, allows delicate and controlled penetration, reducing the risk of tissue damage during manipulation. The purple color offers better visibility on the operating field, facilitating the recognition of the needle between the tissues, a particularly useful element in low light conditions or in complex operations. The length and curvature are calibrated to provide maximum control by the surgeon, both in freehand procedures and when used with suture forceps. The ergonomic design guarantees excellent handling and reduces hand fatigue during prolonged interventions. This needle is packaged under sterile conditions to ensure patient safety and the integrity of the surgical procedure.
The Foqus USP 1 Circular Purple needle is designed to adapt to different types of sutures and can be used effectively both in general surgery and in specialties such as gynaecology, gastroenterology and plastic surgery. The circular shape facilitates homogeneous penetration into the tissue, decreasing local trauma and promoting faster healing thanks to less dispersion of the fibres. The support thread is well aligned to the needle body, offering stability and avoiding unwanted movements during the trajectory of the suture. This instrument meets the highest quality standards required by modern operating rooms, offering safety, resistance and precision in each use.
Therapeutic Indications
Foqus USP 1 Purple Circular Needle is indicated for all surgical procedures that require suturing of soft tissues, such as closing skin incisions, repairing internal lacerations, and tissue reconstruction after trauma or elective operations. Thanks to its versatility, the needle is used both in routine interventions and in highly complex procedures. It is particularly suitable when you want to minimize tissue trauma and maximize the safety and precision of the suture. In abdominal, gynecological or plastic surgery, its homogeneous penetration characteristic reduces operating times and facilitates healing. This needle is mainly chosen where there is a need to preserve tissue structure and promote adequate healing, limiting the formation of scars and wound dehiscences.
The visibility guaranteed by the purple color increases effectiveness even in emergency situations, where speed of use and immediate recognition of the needle are essential. It can be used in combination with various types of suture threads, ensuring optimal compatibility and performance. The circular needle is also indicated for the suturing of delicate and complex tissues, such as blood vessels or parenchymatous organs, where it is essential to maintain a uniform distribution of the penetration force. The use of this instrument helps to reduce post-operative complications due to inadequate suturing techniques, making the procedure safer for the patient and promoting a better quality of the final result.
Composition
The Foqus USP 1 Circular Purple needle is made of special surgical steel, characterized by high mechanical resistance and an excellent ability to maintain sharpness during the entire procedure. The steel undergoes hardening and passivation treatments, guaranteeing the absence of porosity and preventing the risk of releasing particles into the surgical area. The surface is treated so that the needle slides easily through the tissues, minimizing trauma and local coagulation.
The purple color comes from a safe and non-toxic surface treatment, which complies with the requirements of international safety regulations for medical devices. This coating does not alter the mechanical properties of the needle but guarantees visibility and easily distinguishes the device in the operating field. The special geometry of the tip gives self-threading properties and gradual penetration without excessive rubbing or unnatural compression on the tissue. The needle is supplied in a sterile package, generally accompanied by a suture thread of synthetic or natural material, chosen based on surgical needs and the compatibility of the tissue to be sutured.
Contraindications
Foqus USP 1 Purple Circular Needle should not be used in patients who have a known allergy or hypersensitivity to surgical stainless steel or any finishing materials included in the purple surface treatment. The use of this product is not recommended on tissues whose penetration with a circular needle could entail an excessive risk of structural damage, such as some cardiac or nervous tissues or on highly vascularized structures where the suture entails a high risk of haemorrhagic or necrotic damage. The use in children or in patients with particularly delicate conditions must be carefully evaluated by the surgeon in relation to the expected benefit and the possible risks of local trauma or infection. Finally, it is contraindicated if the needle is expected to come into contact with devices or structures that are incompatible with surgical steel, for example in the presence of sensitive metal implants.
Safety of the drug for the species
The safety of Foqus USP 1 Purple Circular Needle comes from the selection of non-toxic materials, the sterilization during the packaging phase and the quality control tests to which each batch is subjected. The surgical steel used is designed to minimize the possibility of the release of metal ions and has a smooth surface which limits the adhesion of microorganisms. The sterility of the package guarantees a significant reduction in the risk of post-operative infections. During use, the needle maintains its structural integrity even under high workloads, avoiding the risk of breakage during suturing even in resistant fabrics.
The purple surface treatment has been tested to ensure the total absence of the release of harmful compounds and guarantees a stable color over time, without alterations or fading that could compromise its recognizability and effectiveness in the operating field. All phases of the production cycle follow international guidelines for the creation of implantable medical devices, guaranteeing the absence of contaminants and ensuring maximum safety for all animal or human target species where permitted. Use is reserved for trained healthcare personnel, reducing the risks associated with incorrect handling of the instrument.
Precautions
When handling surgical needles, it is essential to wear personal protective equipment to prevent accidents and possible cross-contamination. Before use, check the integrity of the sterile package: tampering or accidental opening of the same precludes the safety of the product and requires the replacement of the needle. The healthcare professional should ensure that the size and curvature of the needle are suitable for the type of procedure planned and the characteristics of the patient. Avoid bending, deforming or reusing the needle, as this will compromise its strength and may increase the risk of breakage or unexpected penetration into tissue.
It is good practice to correctly dispose of used needles according to hospital procedures, to avoid accidental needle sticks or contamination between patients. In case of contact with mucous membranes or damaged skin, carefully disinfect the parts involved and, if appropriate, consult the infectious disease control service. It is recommended not to use needles with visible signs of wear, rusty or deformed, to avoid any intra- or post-operative complications. All procedures must be performed in compliance with asepsis and antisepsis guidelines for patient and operator safety.
Method of administration
Foqus USP 1 Purple Circular Needle must only be used by suitable medical or healthcare personnel with experience in surgical suturing techniques. The needle is generally used in combination with a suitable surgical needle holder and compatible threads for the type of operation to be performed. At the end of the preparation phase, the needle is removed from the sterile package using aseptic instruments and inserted into the tissue according to the chosen suturing technique (continuous suture, separate stitches, X-shaped flap, etc.).
The particular curvature of the needle allows you to follow a controlled trajectory, minimizing the risk of damage to the surrounding tissues and facilitating the passage of the thread. During the maneuver, avoid sudden movements or excessive twists which can compromise the stability of the needle on the thread. Once the suture is finished, the needle must be immediately disposed of in the appropriate sharps bins. Correct management of the material and compliance with the technique guarantee the safety and success of the surgical procedure.
Treatment program
The treatment program involving the use of Foqus USP 1 Purple Circular Needle varies based on the type of surgery and the patient's clinical condition. In the case of planned surgical procedures, the surgeon will have to plan the quantity of material needed and evaluate the possibility of replacing the needle during the operation in case of wear or particular tissue resistance. In an emergency setting, the needle can be used immediately, as long as it is available in sterile and integral packaging.
The needle can be used for the immediate closure of surgical incisions or for the creation of deep sutures; in the case of prolonged operations, the sequential use of multiple needles may be necessary to ensure maximum efficiency and integrity of the suture. In the post-operative follow-up, the type of needle used is documented in the medical record, useful for the traceability and management of any complications. In the presence of local infections, the surgeon is required to use only new and sterile needles, avoiding any cross-contamination. Suture planning must take into account the type of tissue, thickness and elasticity, choosing the most suitable combination for each individual patient.
Target species
Foqus USP 1 Purple Circular Needle is intended for use in veterinary and human surgery where protocols allow. It is used on a wide range of animal species, including dogs, cats, exotic animals, livestock and, where compliant with current regulations, in humans. The selection of the device is made based on the anatomical characteristics of the patient, the type of operation and the specific needs of the surgical procedure. In case of use in particular species, a preventive evaluation of the needs for diameter, length and resistance of the needle is recommended to ensure optimal performance and maximum intraoperative safety.